

A devastated pet owner has hit out at the "reckless" administration of the arthritis drug Librela, claiming it has turned her beloved Cavalier King Charles Spaniel's life into one of "emotional turmoil". Rebecca Hudson's 10-year-old dog, Sam, has endured three major surgeries and cost a staggering £50,000 in vet fees since 2021, after she claims the drug was administered by her vet without her consent.
What began as a so-called "wonder drug" treatment for elbow dysplasia quickly spiralled into a series of debilitating side effects, including pancreatitis and stomach upsets, culminating in the rapid destruction of Sam's joints, who was just four years old at the time. The situation reached a crisis point when Sam's ankles swelled so much that it was "like he had had doughnuts inserted under his skin".
"It was really alarming," Ms Hudson told the Express.
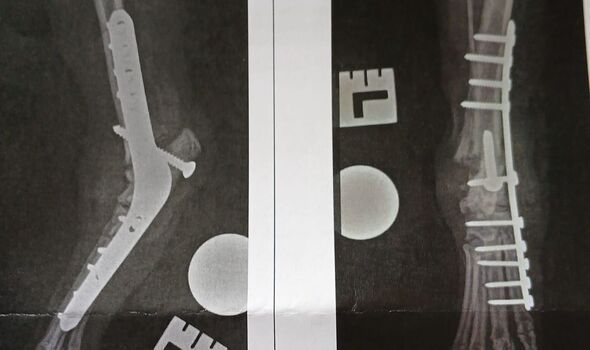
X-rays later revealed that his right ankle was "totally destroyed," requiring a complex arthrodesis surgery - a permanent fusion of the joint using plates, pins and screws. But the nightmare didn't end there. Following a bone graft failure and the subsequent collapse of his left ankle on his 10th birthday, Sam has spent years in recovery.
On Thursday (March 19), the campaign group Librela Solensia The Truth UK sent a damning letter to Health Secretary Wes Streeting, highlighting the "potential risk to human life" associated with Librela and Solensia (for cats). The letter, shared with the Express, raised the alarm over 58 recorded cases of accidental human exposure to Librela - including six instances where vets accidentally injected their own clients.
The group warned that because these are "novel biologics" with no known antidote, the drugs can remain in a human's system for up to 150 days, potentially causing life-threatening reactions like anaphylaxis.
The letter further alleged a "regulatory failure" between the Veterinary Medicines Directorate (VMD) and the Medicines and Healthcare products Regulatory Agency (MHRA), claiming the human health regulator holds "zero recorded cases" - obtained from an FOI - despite the VMD's own data.

Campaigners have called for an immediate suspension of home administration in the UK - a safety measure already adopted in other countries like Australia - warning that the drugs are being stored in domestic fridges and handled by pet owners without adequate safety data.
"We cannot allow [what has happened to pets] to happen to humans," the group urged, noting the drugs' similarities to Tanezumab, a human drug candidate whose trials were halted in 2010 after Pfizer suspended its clinical program at the FDA's request. Trials saw "rapidly progressive osteoarthritis" and even death.
"Two people died during the Tanezumab trials, thought to be unrelated to the drug, but as we have seen from the animal versions, deaths and side effects which the manufacturer thought were unrelated to the drugs are, as proved in real-life use, truly caused by the drugs," the letter reads.
The letter also demanded a formal investigation into the VMD's regulatory oversight and a mandatory update to UK safety data sheets to match the more comprehensive warnings provided to pet owners and clinicians in the US. The US client information sheet explicitly lists serious side effects, including death.
"It appears that the VMD's main priority is to protect the interests of the manufacturer rather than those they are charged to protect, the public and animals," it added.
Ms Hudson's heartbreak is not alone. Debbie Gilbert, a co-founder of Librela Solensia The Truth UK and author of the letter sent to Mr Streeting, lost her two litter sisters, Gem and Chrisse, within just one month of each other. Debbie told the Express that her vets suggested the injections despite both dogs being on other medications - meaning the concomitant use of the drugs was essentially experimental.
"I was not advised of this, or I would never have agreed," Ms Gilbert said, describing a harrowing list of symptoms that followed, including laryngeal paralysis, neurological disorders and dementia-like symptoms. Despite her growing concerns, she claimed her vet dismissed online reports of side effects, telling her "don't believe everything you see online" - a comment which is now documented in her pets' medical notes.
Eventually, both dogs were euthanised due to a complete loss of quality of life. Ms Gilbert is now pursuing a malpractice complaint against seven vets at the practice with the Royal College of Veterinary Surgeons (RCVS).
"The trauma of what they went through haunts me every single day," she said.
While the VMD recently stated that it is "satisfied" that Librela's benefits outweigh its risks for the "vast majority," Ms Hudson is sceptical: "It is irresponsible and unforgivable. People deserve to be given all of the facts before making a decision on whether or not to use this drug".
"I was not asked permission - the jab had been given before we picked him up from his CT scan. I knew nothing of this drug, and nothing about how it works in the body was described to me.
"If I knew then what I know now, I would have never gone near Librela, and I would also have reported the vets to the RCVS immediately for administering a drug without my consent."
The specialist orthopaedic professor who performed Sam's surgeries expressed a chilling "suspicion" regarding the drug's role in the spaniel's decline. In an email sent to Ms Hudson, shared with the Express, the professor revealed a "growing concern" among global orthopaedic specialists that Librela may be linked to rapid-onset arthritis, severe joint instability, or even total joint destruction in some dogs.
While the link remains unproven, the surgeon admitted he had no other "good explanation" for why Sam's ankles had simply collapsed: "If there is a link between Librela and these conditions, the cause or reason is as yet unknown and unexplained," he wrote.

The Express contacted the VMD and the MHRA for comment. A spokesperson for the VMD said: "All veterinary medicines undergo rigorous scientific assessments, including human safety, prior to approval and are constantly reviewed to ensure the benefits of use of the product remain positive.
"The exposure to humans during the administration of Librela is very uncommon, less than 1 case per 10,000 estimated animals treated; and in order for pet owners to administer the products to their pet at home, the prescribing vet must be confident they are competent to do so."
The MHRA declined to comment.